Now, the contents of this journal article will not be news to most in this community; however, it signals a pivotal step towards mainstream recognition of CVT from the mRNA vaccines. Thus, I find myself in the position of wanting to raise your optimism while tempering expectations.
What is the study not?
This study says nothing about a causal link between mRNA vaccines and CVT. Nor does it say anything about the damage the vaccines have potentially caused or the rate at which the people who receive the vaccine may be afflicted with CVT. Furthermore, despite the fact that the study covers both mRNA vaccines and an adenovirus vaccine, one must be careful when interpreting comparisons between these three vaccines.
There are a few barriers to the methodology the authors used (disproportionality analysis) that we must always keep at the forefront of our minds. First of all, we do not have good information on the denominators of adverse event reports. In other words, we do not know how many vaccinations were administered. Secondly, this is confounded by the fact that there may be asymmetric reporting of adverse events depending on providers. Some hospitals or clinics, for example, may not report any adverse events and some may report every single one. This is exactly the same problem that we run across with VAERS and is, in fact, the reason most reasonable people point to VAERS as vastly under-reporting the incidence of adverse events (rather than over-reporting). Finally, there may be additional confounding variables, ie., drug to drug interactions.
Additionally, and this is even harder to quantify, if there is a vaccine or drug that causes a huge range of adverse events, this may actually make it more difficult to spot a singular adverse event like CVT. If, for example, a drug causes CVT and not much else, then the proportion of this type of adverse events within the total of all adverse events for that particular drug will be high and it will be easy to spot when comparing the drug to the database at large. However, mRNA vaccines cause many events, which, in a way, waters down the proportion of CVT within the drug.
I hope that last point was clear, but if it was not, please refer to the example table below to get a picture of how this is significant and don’t hesitate to ask for clarification!
What is the study?
Disproportionality analysis essentially searches through drug reaction databases and looks for differences in the proportions of certain types of adverse events by comparing the drug to, generally, all drugs in the database. The reasoning is that even if there are some events unrelated to the drug, these events should happen more or less at random, and, thus, be more or less equally likely to occur in any given drug.
I will use an analogy — but note, this is just an analogy and no analogy is perfect.
If you were to sample water from ditches near a bunch of farms, you would expect to find pesticides in each ditch. The pesticide level would be quite low at, maybe, a few parts per million. If, however, the pesticides are high in one ditch, then you would have a disproportionality (in comparison to the other ditches). Yet, there is no causal link because it does not tell us why the pesticides are higher in this ditch. Did that farm spray more than the recommended level of pesticides, was there a recent runoff from rainfall into the ditch, or did something else happen entirely? Unclear.
What disproportionality analysis tells us is that there is something there in a higher proportion than one would expect to see there. In other words, it is a “hypothesis generation” rather than a “hypothesis testing” methodology. There is, then, one more step that needs to be taken following disproportionality analysis and that is to actually initiate an investigation into the individual cases to find whether or not there is a causal link.
That is why I want to temper expectations: regulators have shown little curiosity to investigate these links except in the case of the adenovirus vaccines. Western scientists are even more afraid than the regulators and put on their horse blinders whenever possible. It should be noted, even this study came out of South Korea, where perhaps not the level, but the kind of regulatory and scientific capture is distinctly different than in the west.
Disproportionality analysis is an excellent ‘canary in a coalmine’ technique in order to entice future research. This technique is not innocuous and has been used to identify adverse events in the past, which has lead to many drugs being taken off the market.
Preamble over: let’s get to the study
So, this study essentially looks at both mRNA vaccines and the Astrazeneca vaccine. As one would expect for a vaccine where a causal link has already been established, the authors did find safety signals for the Astrazeneca vaccine. However, what the authors are more interested in is the fact that they found significant safety signals for both mRNA vaccines as well. Albeit, there are curious differences between the two safety signals.
Recall that I said we must be careful in interpreting comparisons between the vaccines? Well, I am just going to downplay the underlying rates for that reason.
As we can see, there are clear differences. There are more events with Astrazeneca, more deaths, and a higher rate of seriousness. Though, there is also a higher rate of full recovery without sequelae from Astrazeneca. Frankly, none of that is not interesting to me. Reporting with Astrazeneca should be more homogeneous due to where the doses have been administered and one would expect CVT to be more carefully monitored in a vaccine where a causal link has already been found. There is also more institutional bias in favor of the mRNA vaccines that would likely prevent most reports of such an event from the vaccine.
But the authors point out an interesting finding. A finding that would be completely counterintuitive if there was no there there:
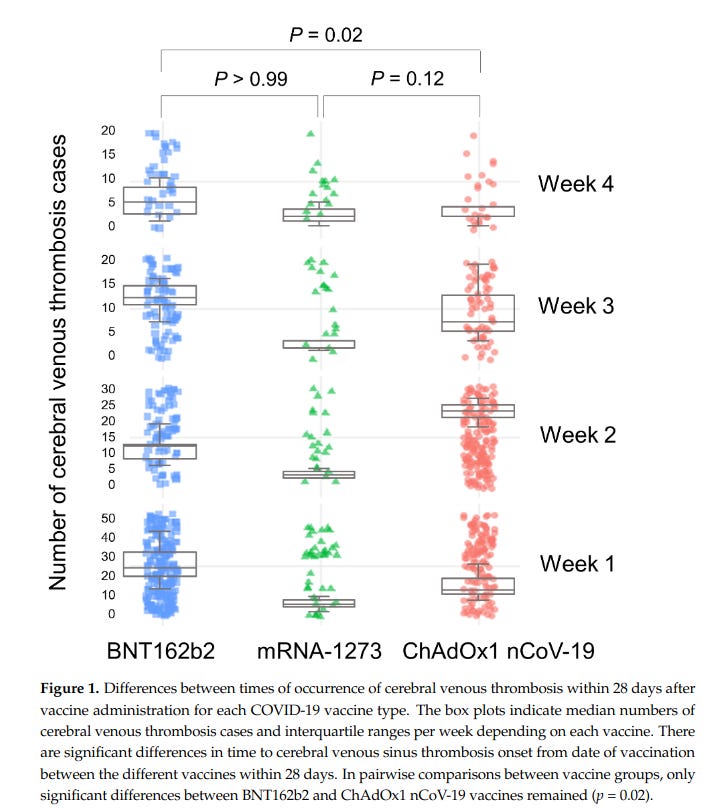
The mRNA-based COVID-19 vaccines had the highest number of CVT cases in the first week after vaccination, after which the incidence decreased gradually. In contrast, the ChAdOx1 nCoV-19 vaccine showed the highest incidence of CVT in the second week after vaccination and a sharp decrease thereafter. These timelines suggest that the thrombosis mechanisms of these vaccines differ.
And here is a graphical representation of that finding:
What is going on there? The authors go so far as to generate a plausible hypothesis… and it presents a scary scenario for those that took the mRNA vaccines.
The mRNA-based COVID-19 vaccines . . . directly induce intracellular production of the spike protein . . . This evidence supports a wide temporal distribution of CVT occurrence related to mRNA-based COVID-19 vaccination in our study. In contrast, the formation of PF4 antibodies and PF4–polyanion complexes in vaccine-induced immune thrombotic thrombocytopenia, the most well-known thrombotic complication after ChAdOx1 nCoV-19 vaccination, takes time. A type-II heparin-induced thrombocytopenia that develops via a similar mechanism occurs 5 to 14 days after exposure due to the time required for the formation of antibodies.
This is damning if true because these vaccines are not just generating antibodies, but they are making people into spike protein factories for months on end. If CVT is being caused by the spike proteins (which is an obvious implication), then it may be up to six months before individuals who took the mRNA vaccines are no longer at risk of CVT (at a rate higher than base). Whereas, for Astrazeneca, if someone does not have a CVT event in the first month or so, they may be in the clear.
Furthermore, there is no reason to believe that this is unique to CVT. We would expect this to occur for most adverse events with the mRNA vaccines.
Yikes!







This would seem to tie into today's Substack by Walter Chestnut, Absolute Confirmation of my Spike Protein Amyloidosis Hypothesis:
https://wmcresearch.substack.com/p/absolute-confirmation-of-my-spike?s=r